 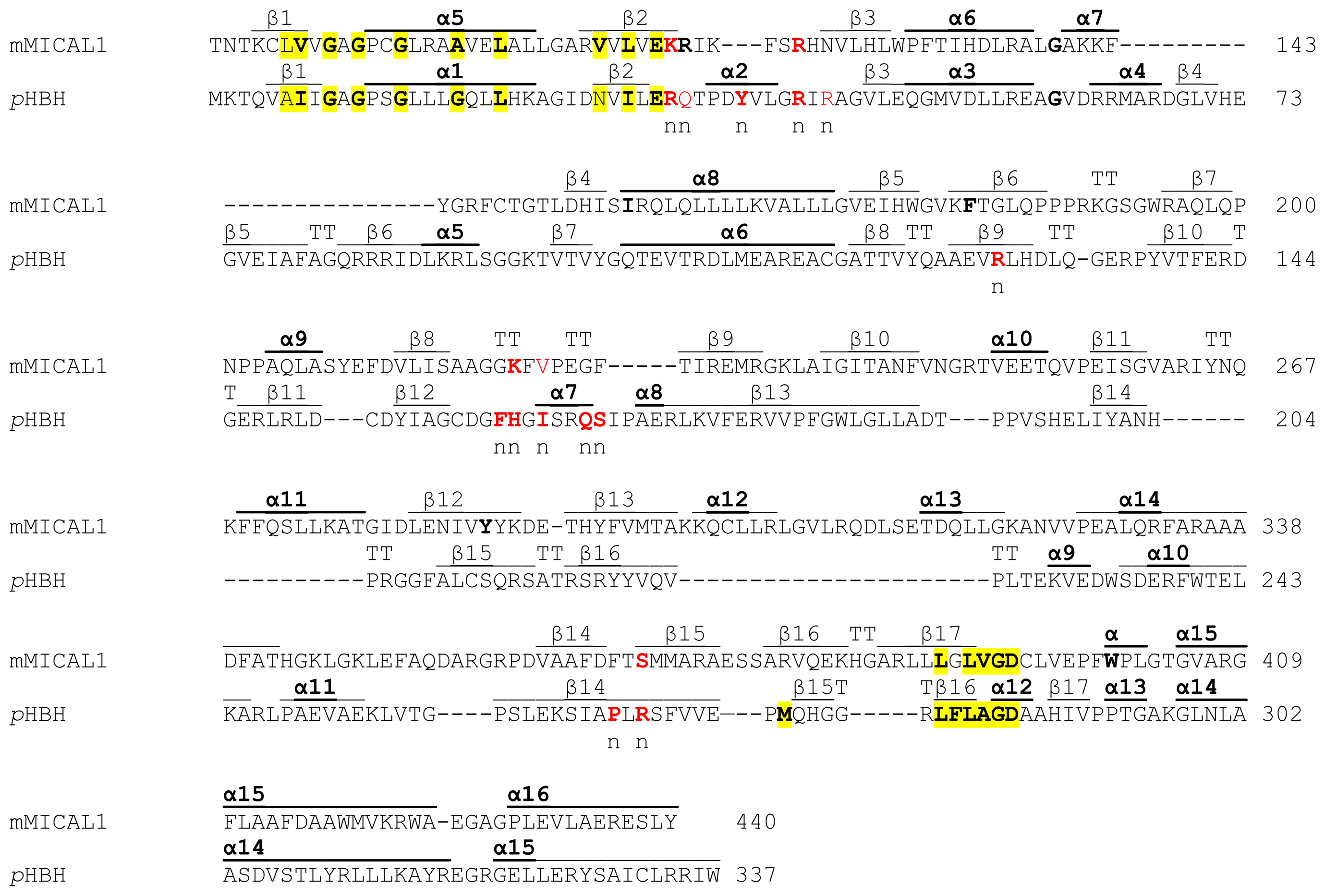
New methods for improved biliverdin bioavailability are necessary to develop improved bright far-red and near-infrared fluorescent proteins for noninvasive imaging in vivo. However, the availability of biliverdin limits the fluorescence of biliverdin-attaching fluorescent proteins hence, extra biliverdin is needed to enhance brightness. Novel applications of smURFP include in vitro protein diagnostics with attomolar (10−18 M) sensitivity, encapsulation in viral particles, and fluorescent protein nanoparticles. 
The small Ultra-Red Fluorescent Protein is biophysically as bright as the enhanced green fluorescent protein, is exceptionally photostable, used for biosensor development, and visible in living mice. The small Ultra-Red Fluorescent Protein (smURFP) was evolved from a cyanobacterial phycobiliprotein to covalently attach biliverdin as an exogenous fluorophore. New fluorescent proteins based on phytochrome and phycobiliproteins use biliverdin IXα as fluorophores, do not require oxygen for maturation to image anaerobic organisms and tumor core, and do not generate hydrogen peroxide. Coral-derived red fluorescent proteins require oxygen for fluorophore formation and release two hydrogen peroxide molecules. The ideal fluorescent proteins are bright, photobleach minimally, express well in the desired cells, do not oligomerize, and generate or incorporate exogenous fluorophores efficiently. Far-red and near-infrared fluorescent proteins absorb light >600 nm to expand the color palette for imaging multiple biosensors and noninvasive in vivo imaging. Near-infrared light penetrates biological tissue with blood vessels due to low absorbance, scattering, and reflection of light and has a greater signal-to-noise due to less autofluorescence. We detail the continuous transformation between interwoven molecular and electrical information over long timescales in this system, showing how biochemical activity, invisible to most large scale recording techniques, is the key computational currency directing a life-or-death decision.Noninvasive fluorescent imaging requires far-red and near-infrared fluorescent proteins for deeper imaging. The eruption functions like an action potential at the level of the network, transforming deliberation within the network into an all-or-nothing output, after which the male will no longer sacrifice his life to continue mating. When consensus is reached, PKA pushes the network toward a large-scale and synchronized burst of calcium influx, which we call an eruption. Protein Kinase A (PKA) activity contrasts this internal measurement with input from the other neurons to represent evidence that the network’s goal has been achieved. Each neuron in a recurrent network measuring time into mating contains slightly different internal molecular estimates of elapsed time. Challenging this view, we show that a male fly’s decision to persist in mating, even through a potentially lethal threat, hinges on biochemical computations that enable processing over minutes to hours. Computations in the brain are broadly assumed to emerge from patterns of fast electrical activity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
